Photo credits: ScenTree SAS
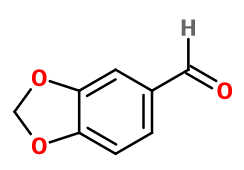
Heliotropin
1,3-benzodioxole-5-carbaldehyde ; 3,4-dihydroxybenzaldehyde methylene ketal ; 3,4-dimethylene dioxybenzaldehyde ; Dioxymethylene procatechuic aldehyde ; Dioxymethylene protocatechuic aldehyde ; 5-formyl-1,3-benzodioxole ; Geliotropin ; Heliobase ; 3,4-methylene dioxy benzaldehyde ; 3,4- methylene dioxybenzaldehyde ; 3,4- methylenedioxybenzaldehyde ; Piperonal ; Piperonaldehyde ; Piperonyl aldehyde

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | Purity | Latin name | Treated part | Geographical origin | MOQ |
|---|---|---|---|---|---|---|---|---|---|---|
|
|
Heliotropine - 30 Gr | - |
Visit website
|
- | - | - | - | - | - |
General Presentation
-
CAS N° : 120-57-0
-
EINECS number : 204-409-7
-
FEMA number : 2911
-
FLAVIS number : 05.016
-
JECFA number : 896
-
Appearance : White solid
-
Density : 1,34
-
Volatility : Base
-
Price Range : €€
Physico-chemical properties
-
Molecular formula : C8H6O3
-
Molecular Weight : 150,13 g/mol
-
Log P : 1,05
-
Fusion Point : 37°C
-
Boiling Point : 264°C
-
Detection Threshold : 62 ppb et 1 ppm (0,0001%)
-
Optical rotation : Donnée indisponible
-
Vapor pressure : Donnée indisponible
-
Refractive Index @20°C : Donnée indisponible
-
Acid Value : Donnée indisponible.
-
Flash Point : >113°C
Uses
Uses in perfumery :
Heliotropin is used in carnation, mimosa, tuberose, frangipani, ambery and fougere notes. Stable in soap bases, but may be responsible for coloration in cosmetic products. Tends to be gradually replaced by Heliotropex® (base), because heliotropin is regulated on importation beyond certain quantities.
Year of discovery :
Discovered in 1869.
Natural availability :
Heliotropin is present in trace amounts in a natural state in Vanilla Bourbon Absolute, from which it can be extracted.
Isomerism :
Heliotropin does not have any isomer used in perfumery.
Synthesis precursor :
Heliotropin is stored in lockers, as it is a precursor for the synthesis of MDA (methylenedioxyamphetamine).
Synthesis route :
Heliotropin can be synthesized in several ways. A first method starts with Isosafrole (derived from Safrole, isolable from various essential oils) as it is capable of undergoing an oxidation with chromium salts and oxygen or ozone, to obtain Heliotropin. A second synthetic route uses Catechol, converted to 3,4-dihydroxymandelic acid by reaction with glyoxylic acid, in the presence of aluminium oxide. An oxidation followed by a decarboxylation allows to obtain a final intermediate product, capable of undergoing a reaction with dichloromethane in order to form the acetal function of the molecule (among other things), in the presence of quaternary ammonium salts.
Regulations & IFRA
Allergens :
This ingredient does not contain any allergen.
IFRA 51th :
This ingredient is not restricted for the 51th amendment

